Trailblazing innovation keeps donated human eyes healthy for longer
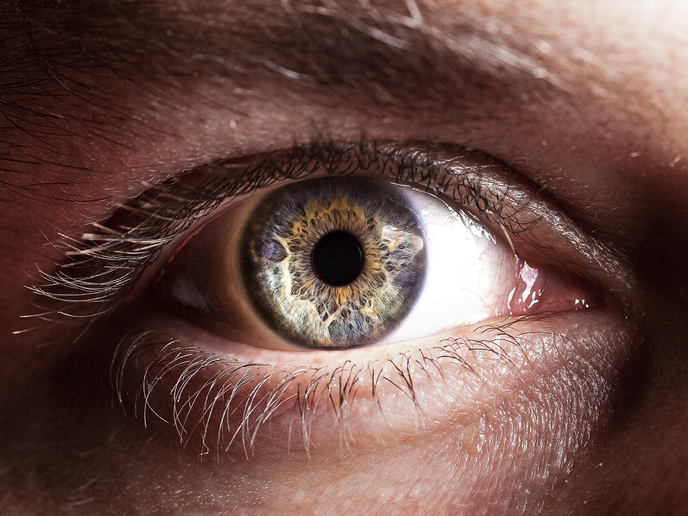
A lack of donated human organs and the difficulty in preserving them is holding back the development of regenerative therapies and new drugs. A new approach to reviving and maintaining donated human eyes is aiming to get past this barrier, opening up new possibilities for treatment of retinal degeneration and many other conditions that can lead to impaired vision and blindness. Pia Cosma led the ECaBox(opens in new window) project, at the Centre for Genomic Regulation(opens in new window) (CRG) in Barcelona, where a multidisciplinary team is creating a device that resuscitates eyes from the dead. “We are aiming to enable preclinical testing in whole eyes. Donated human eyes undergo fast damage, in a few hours, as soon as blood perfusion ceases. ECaBox allows for resuscitation and preservation outside the body,” explains Cosma, ICREA research professor(opens in new window) and group leader at the CRG, and coordinator of the project. ECaBox can be used to preserve eyes that can then be used to test therapies for glaucoma, age-related macular degeneration, dry eye, diabetic retinopathy, retinitis pigmentosa and many more. As Cosma adds: “An eye preserved outside of the body could be used for research for almost any eye disease and the application of a therapy to any compartment of the eye, for any disease, could be studied.”
Innovative procedures keep post-mortem eyes perfused and healthy
While advances in human organoids, tissues grown in a Petri dish, are improving how the function of an eye is mimicked, they fail to encapsulate the eye’s physiological complexity. As organoids typically do not have blood vessels, they cannot correctly model the complexity of the human retina, which relies on nutrients delivered through blood vessels. “Any retinal therapy acts in this vascular-neuronal dynamic, so it is important to understand how therapies work in that physiological context before testing in humans,” says Cosma. By using software and 3D design, the team designed the ECaBox to support donated eyes, and hold a perfusion tube in place so fluid flow could be restored in the eye’s blood vessels. Human donor eyes were collected after death with written consent obtained from family members, following Spanish law and approval from hospital ethics committees. “Pumping and oxygenation systems control fluid flow and corneal hydration while eyes are maintained outside the body,” notes Cosma. To resuscitate and maintain the eyes, a thin tube is inserted into an eye artery and secured in place. Electrophysiology perfusate, a specialised, controlled saline or buffer solution, is pumped through the artery at controlled flow rates and eye structure and blood vessel filling examined using retinal imaging, optical scans and high-resolution 3D X-ray imaging. The images are processed, using artificial intelligence (AI) models to identify and analyse blood vessels. Vessel networks are aligned, divided into regions and measured for length, volume and density to allow comparison between eyes. AI is also playing a key role: “We can determine retinal health using tissue staining, cell imaging, flow cytometry to measure live cells, and electrical recordings of retinal responses to light. Retinal damage can be graded and then classified using AI models trained on histology images.”
Important step forward in the field of organ transplantation
The study is a proof of concept showing that ex vivo preserved organs are useful in therapy development and also has implications in the organ transplant field. ECaBox will greatly reduce pharma costs, enabling the testing of many more therapies. “Indeed, we have received interest from the USA and the data we have provides strong evidence that preclinical tests can be evaluated in post-mortem tissues, as part of the standard battery of preclinical tests a pharma company would perform with a new therapy,” Cosma adds.